CK-MB Isoenzyme MB of Creatine Kinase test kit
1. INTEDED USE
This kit is applicable to the in vitro quantitative detection on the content of Creatine Kinase-MB (CK-MB) in human serum/plasma/whole blood samples and is mainly used for implementing auxiliary diagnosis of acute myocardial infarction. This kit only provides test result of Creatine Kinase-MB (CK-MB). The obtained result should be analyzed in combination with other clinical information.
2. PRODUCT SPECIFICATION
| Model No. | CK-MB |
| Methodology | Fluorescence Immunochromatographic Assay |
| Sample Type | serum/Plasma/Whole Blood |
| Time to Result | 10-15mins |
| Storage | 2~30 ℃/36~86℉ |
| Shelf Life | 24 months |
| Certificate | ISO13485,CE,MHRA |

| MAIN KIT COMPONENTS
* Test device * Sample diluents * Instructions for Use |
PACKING
* 25 test /kit * Aluminum foil bag labeling * shrink wrap |
3.TEST METHOD
4.CLINICAL PERFORMANCE
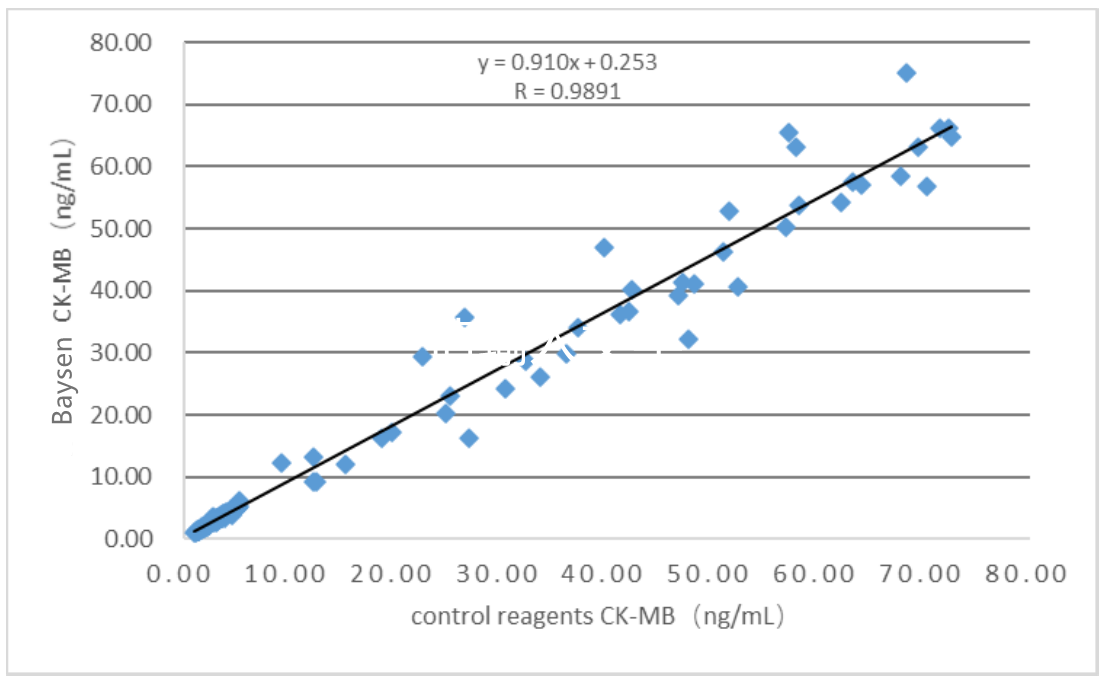
Clinical evaluation performance of the product is assessed through collecting 186 clinical samples. The results are compared by using the corresponding kit of chemiluminescence as the control reagent. Their comparability is studied by linear regression. The correlation coefficients of the two tests are Y=0.910X+0.253 and R=0.9891, respectively.
5. CERTIFICATE
* ISO System Certificate
* CE Certificate
* EU Registration
* UCKA MHRA Registration








