Early Pregnancy Test HCG Human Chorionic Gonadotropin urine rapid test
1. INTEDED USE
This kit is applicable to in vitro qualitative detection of human chorionic gonadotropin (HCG) in serum sample,which’s suitable for auxiliary diagnosis of early trimester of pregnancy. This kit only provides human chorionic gonadotropin test results, and results obtained shall be used in combination with other clinical information for analysis.
2. PRODUCT SPECIFICATION
| Model No. | HCG |
| Methodology | Colloidal Gold |
| Sample Type | Urine/Serum |
| Time to Result | 10-15mins |
| Storage | 2~30 ℃/36~86℉ |
| Shelf Life | 24 months |
| Certificate | ISO13485, CE Certificate, UCKA MHRA Certificate |

|
MAIN KIT COMPONENTS * Test device * Sample collection tube * Disposable pipette * Instructions for Use |
PACKING * 25 test /kit * Aluminum foil bag labeling * shrink wrap
|
3.TEST METHOD
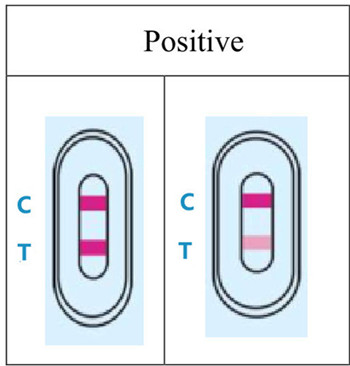
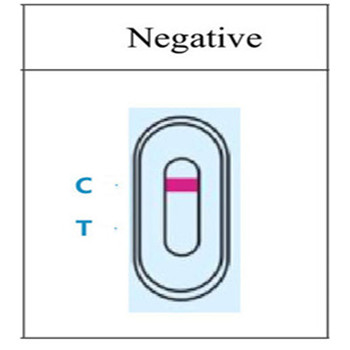
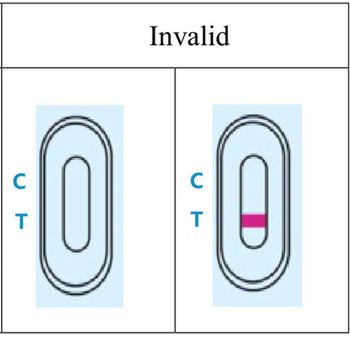
4.RESULT EVALUATION AND EXPLANATION
5.CLINICAL PERFORMANCE
Clinical performance of this product’s assessed through collection of 311 cases of clinical samples. Marketed kit of colloidal gold’s used as reference reagent. The BAYSEN reagent test will be compared with the control reagent:
| Baysen Result of RV/AV |
Test result of Reference reagent | Positive coincidence rate: 99.40% (95%C.I. 96.69%~99.89%) Negative coincidence rate: 100.00% (95%C.I.97.40%~100.00%) Total coincidence rate: 99.68% (95%C.I.98.20%~99.94%) |
||
| Positive | Negative | Total | ||
| Positive | 166 | 0 | 166 | |
| Negative | 1 | 144 | 145 | |
| Total | 167 | 144 | 311 | |
6. CERTIFICATE
* ISO System Certificate
* CE Certificate
* EU Registration
* UCKA MHRA Registration