Fecal Occult Blood Fit Colon Cancer Bowel Cancer Screening Rapid Test
1. INTEDED USE
This kit is used for qualitative detection of hemoglobin in human stool sample and is used for evaluating of gastrointestinal bleeding. The kit only provides the test result of f hemoglobin, and the obtained result shall be analyzed in combination with other clinical information. It must only be used by healthcare professionals.
2. PRODUCT SPECIFICATION
| Model No. | FOB |
| Methodology | Colloidal Gold |
| Sample Type | Faces |
| Time to Result | 10-15mins |
| Storage | 2~30 ℃/36~86℉ |
| Shelf Life | 24 months |
| Certificate | ISO13485, CE Certificate, UCKA MHRA Certificate |

|
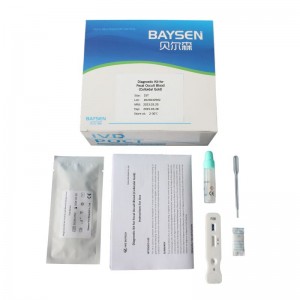
MAIN KIT COMPONENTS * Test device * Sample collection tube * Disposable pipette * Instructions for Use |
PACKING * 20 test /kit * Aluminum foil bag labeling * shrink wrap
|
3.TEST METHOD
| 1 | Use sample collection tube to collect, mix well and dilute the sample. Use sampling stick to take about 30mg of stool. Then, transfer the stool to a sample collection tube containing sample diluent, tighten by rotating, and shake sufficiently. |
| 2 | If stool of patient with diarrhea is loose, use disposable pipette to draw the sample, add 3 drops (about 100μL) of sample-to-sample collection tube, and shake the sample and sample diluent sufficiently. |
| 3 | Take the test device out of the aluminum foil bag, place it on a horizontal worktable flat, and make a proper mark. |
| 4 | Discard the first two drops of diluted sample. Then, vertically, and slowly add 3 drops (about 100μL) of bubble-free diluted sample to the center of sample hole of the test device and start timing. |
| 5 | The result shall be read within 10-15 minutes. Test result obtained after 15 minutes is invalid (for detail about result see Interpretation of Test Results). |
4.RESULT EVALUATION AND EXPLANATION
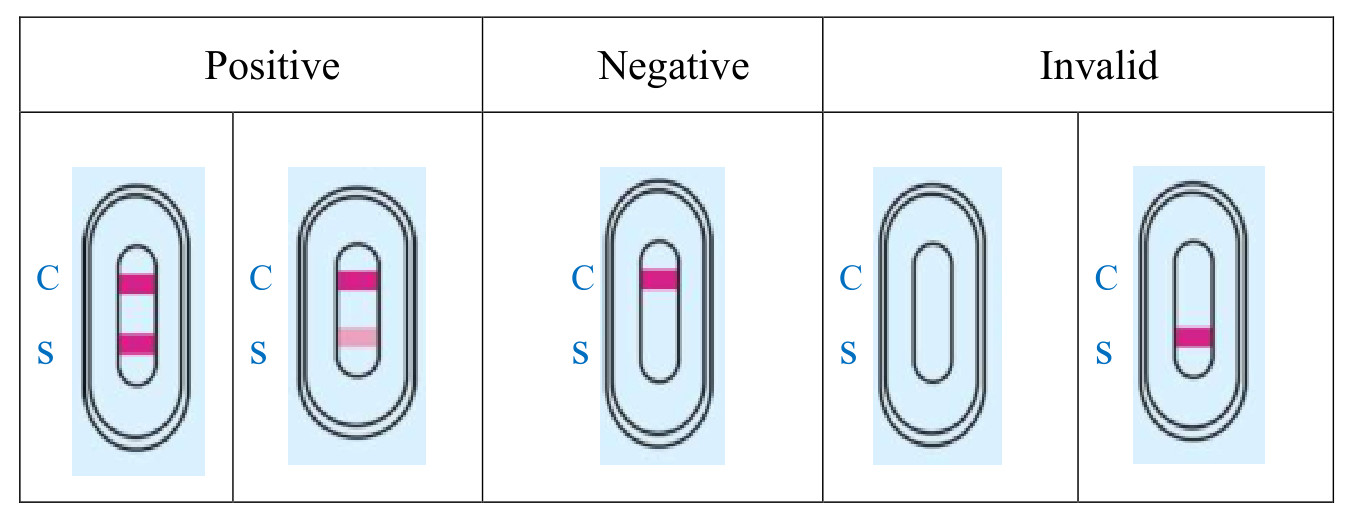
5.CLINICAL PERFORMANCE
Clinical evaluation performance of the product is assessed by collecting 288 clinical samples. Sample donors include patients with gastrointestinal bleeding and normal people. The corresponding kit of colloidal gold method is used as the control reagent. The WIZ BIOTECH reagent test will be compared with the control reagent:
| Baysen Result of FOB |
Test result of Reference reagent | Positive coincidence rate:99.33% (95%C.I. 96.32%~99.88%) Negative coincidence rate:100.00% (95%C.I.97.29%~100.00%) Total coincidence rate:99.65% (95%C.I.98.06%~99.94%) |
||
| Positive | Negative | Total | ||
| Positive | 149 | 0 | 149 | |
| Negative | 1 | 138 | 139 | |
| Total | 150 | 138 | 288 | |
6. CERTIFICATE
* ISO System Certificate
* CE Certificate
* EU Registration
* UCKA MHRA Registration