Introduction
Pregnancy testing has evolved dramatically since its inception, transitioning from rudimentary methods to highly sensitive, rapid, and accessible diagnostic tools. Today, the global pregnancy testing market is valued at over $1.3 billion and is projected to grow at a compound annual growth rate (CAGR) of 5.2% through 2030, driven by technological advancements, increasing awareness of reproductive health, and rising demand for home-based diagnostics. This article explores the scientific principles behind modern pregnancy tests, emerging innovations, market dynamics, and ethical debates shaping the field.
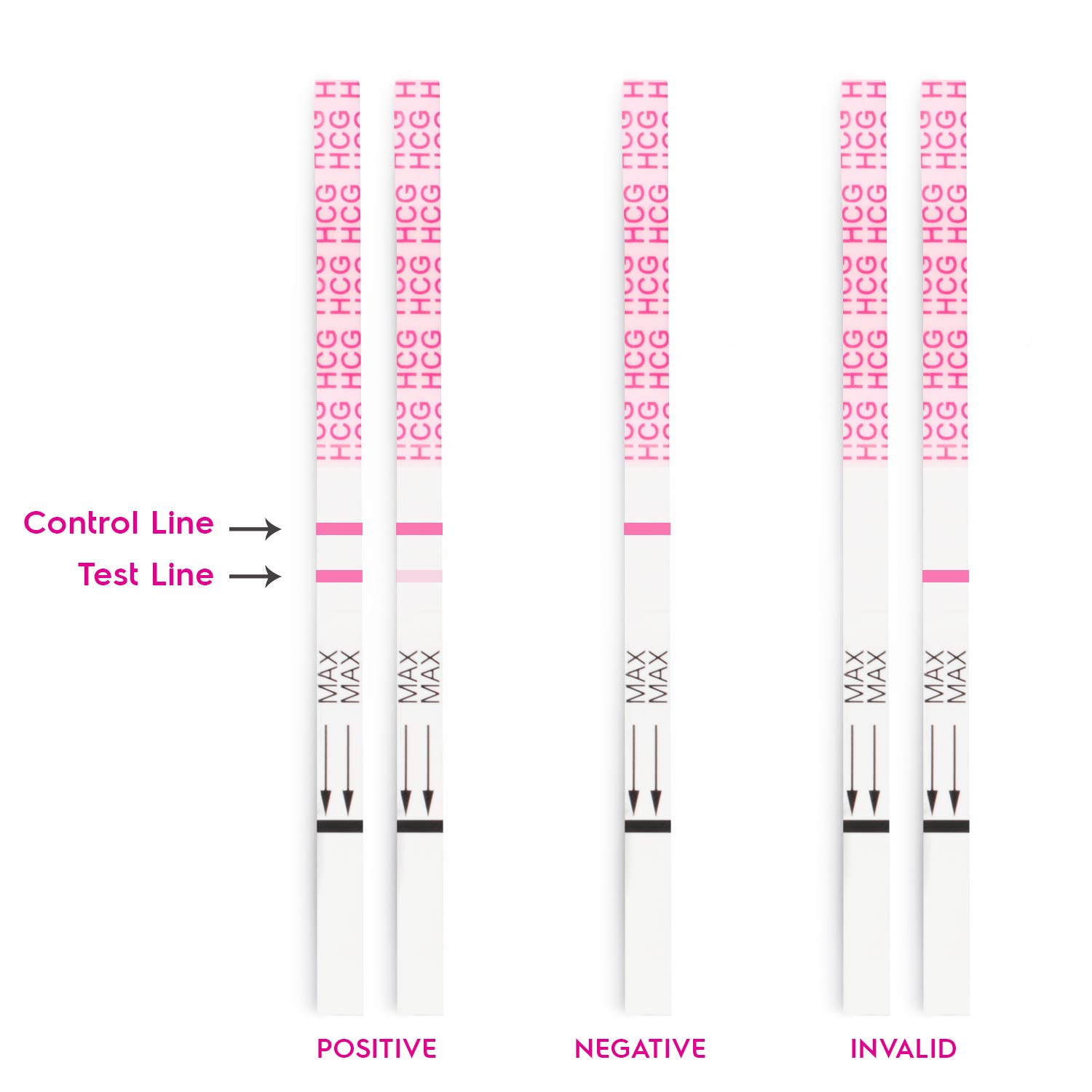
Scientific Foundations of Pregnancy Detection
At the core of pregnancy testing lies the detection of human chorionic gonadotropin (hCG), a glycoprotein hormone produced by the placenta shortly after embryo implantation. hCG levels double approximately every 48 hours in early pregnancy, peaking at 8–11 weeks of gestation.
1. Types of Tests
- Urine-Based Immunoassays**: Over-the-counter (OTC) home pregnancy tests (HPTs) utilize monoclonal antibodies to detect hCG in urine, with sensitivities as low as 10–25 mIU/mL. These lateral flow assays provide results in 3–5 minutes.
- Serum Quantitative hCG Tests**: Conducted in clinical labs, these tests measure hCG levels with greater precision (detecting ≤5 mIU/mL) and are critical for monitoring ectopic pregnancies or gestational trophoblastic diseases.
- Digital and Connected Devices**: Next-gen HPTs integrate Bluetooth-enabled sensors and smartphone apps to quantify hCG levels and track gestational timelines.
2. Accuracy and Limitations
While modern HPTs boast >99% accuracy when used correctly, false negatives may occur due to testing too early (before hCG reaches detectable thresholds) or diluted urine. False positives, though rare, can stem from medications containing hCG (e.g., fertility treatments) or medical conditions like pituitary tumors.
Technological Innovations Reshaping the Market
The pregnancy testing industry is undergoing a paradigm shift, fueled by interdisciplinary advancements in biotechnology, data science, and consumer healthcare.
1. Non-Invasive Prenatal Testing (NIPT) Integration
Emerging products combine pregnancy confirmation with early genetic screening. For instance, some companies now offer “prenatal wellness kits” that detect hCG while analyzing fetal DNA in maternal blood for chromosomal abnormalities like trisomy 21.
2. AI-Powered Diagnostics
Startups are leveraging machine learning to enhance test interpretation. Apps like *Glow* and *Flo* sync with HPTs to analyze result lines under varying lighting conditions, reducing user error.
3. Sustainability Initiatives
To address environmental concerns, brands like *Lia* and *Biodegradable Pregnancy Test* have introduced flushable, plant-based test strips that decompose within 10 weeks.
**4. Multiplex Biomarker Panels**
Researchers at Stanford University recently developed a paper-based test that simultaneously detects hCG, progesterone, and estradiol to assess pregnancy viability, potentially reducing unnecessary clinical visits.
Market Dynamics and Regional Trends
According to Grand View Research, North America dominates 38% of the global pregnancy test market, attributed to high healthcare expenditure and widespread OTC availability. However, Asia-Pacific is poised for the fastest growth (6.8% CAGR), driven by improving healthcare infrastructure and rising female workforce participation in countries like India and China.
Key Players and Competition
- SPD Swiss Precision Diagnostics (maker of Clearblue) leads innovation with products like the *Weeks Estimator*, which predicts conception dates.
- Church & Dwight First Response) and Abbott Laboratories are investing in telehealth integrations, allowing users to share results directly with physicians.
- Emerging competitors like Modern Fertility and Stix target millennials via direct-to-consumer models and subscription services.
Regulatory Landscape
The U.S. FDA classifies HPTs as Class II medical devices requiring premarket notification (510(k)). In 2023, the agency tightened labeling requirements to standardize sensitivity claims and improve user instructions. The EU’s Medical Device Regulation (MDR) now mandates post-market surveillance for pregnancy tests sold in member states.
Ethical and Societal Challenges
While pregnancy tests empower individuals with reproductive autonomy, they also spark complex debates:
1. Privacy Concerns**
Connected devices raise questions about data security, particularly in regions with restrictive abortion laws. In 2022, a U.S. state subpoenaed period-tracking app data in a controversial abortion case, prompting companies like *FemTech* to adopt end-to-end encryption.
**2. Accessibility Disparities**
Despite HPTs costing as little as $0.50 to produce, pricing remains prohibitive in low-income regions. WHO reports that 23% of women in sub-Saharan Africa lack access to reliable pregnancy tests, contributing to delayed prenatal care.
3. Ethical Use in Crisis Settings
Humanitarian organizations like Doctors Without Borders have criticized the use of mandatory pregnancy testing for refugee women, citing violations of bodily autonomy.
4. Over-the-Counter Availability vs. Clinical Guidance**
While HPTs democratize early detection, some clinicians argue that false reassurance from negative results may delay care for ectopic pregnancies or miscarriages.
Future Directions
The next frontier in pregnancy testing lies in multi-functional, AI-integrated platforms. Researchers at MIT are developing a saliva-based nanosensor that detects hCG alongside nutritional deficiencies like folate levels. Meanwhile, advancements in molecular diagnostics may enable conception detection within hours of implantation via CRISPR-based assays.
Personalized Predictive Analytics
Companies like *Aevi* are piloting tests that combine hCG data with menstrual history and biomarkers to predict due dates and miscarriage risks, potentially integrating with electronic health records (EHRs).
Policy Recommendations
Experts urge governments to:
- Subsidize HPTs in low-resource settings
- Enact stricter data privacy laws for femtech devices
- Standardize global sensitivity thresholds for OTC tests
Conclusion
Pregnancy testing has transcended its role as a mere diagnostic tool, becoming a nexus of technological innovation, public health strategy, and ethical discourse. As science continues to push the boundaries of early detection, stakeholders must balance commercial interests with equitable access and privacy protections. With the global fertility rate fluctuating and reproductive rights under scrutiny, the humble pregnancy test remains a critical—and evolving—component of modern healthcare.
Post time: Mar-17-2025